 When the temperature increases along the heat input, the entropy also increases, but the example of a process without change of temperature is more clear in my opinion. The faster-moving particles have more disorder than particles that are moving slowly at a lower temperature. An increase in temperature means that the particles of the substance have greater kinetic energy. P3A5A heat engine does work as a result of. So, the denominator increased, but the numerator increased much more. Entropy increases as temperature increases. TOPIC 3B Entropy changes accompanying specific processes TOPIC 3E Combining the First and Second Laws FOCUS 4. 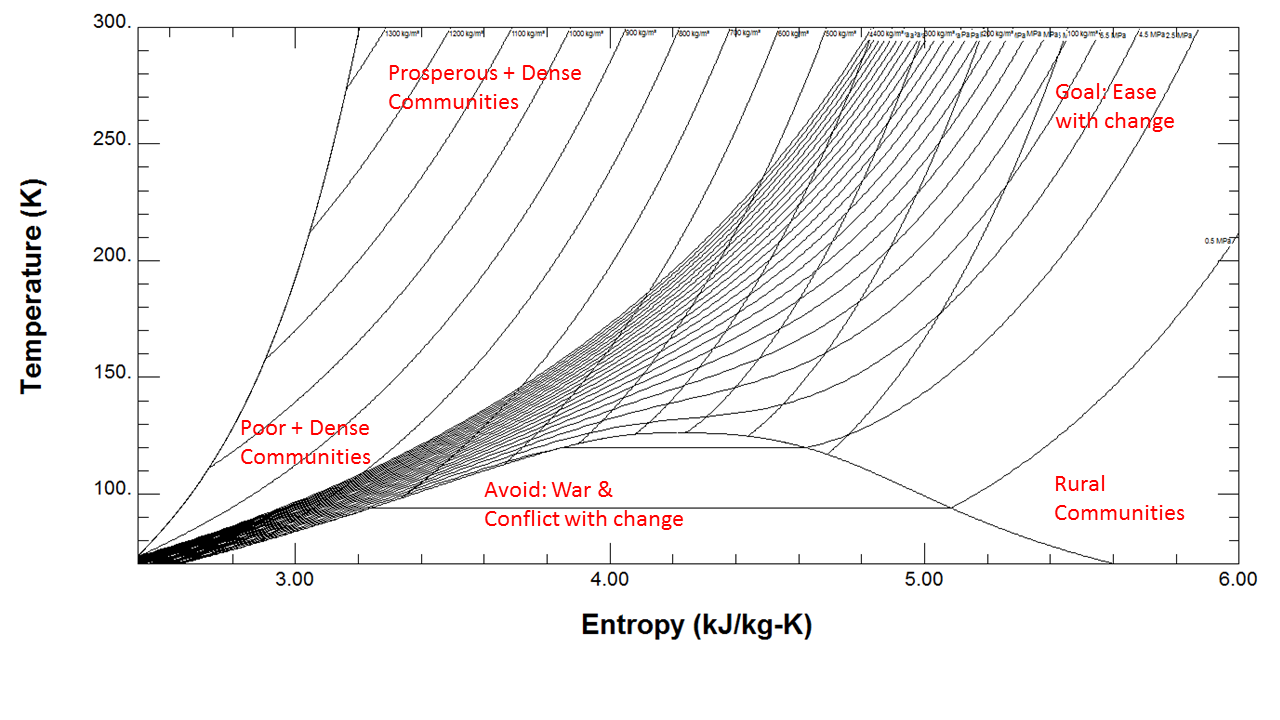
A differential change in entropy is defined by Entropy is one of the few quantities in the physical sciences that require a particular direction for time, sometimes called an arrow of time.As one goes 'forward' in time, the second law of thermodynamics says, the entropy of an isolated system can increase, but not decrease. The materials high-temperature phase thus. 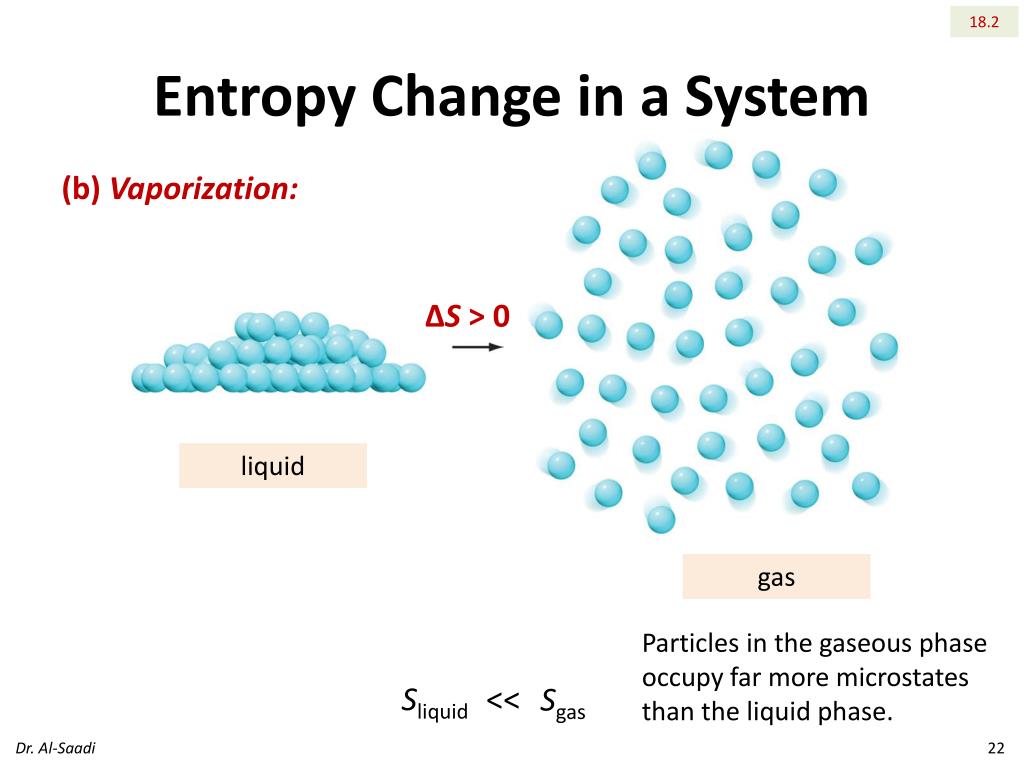
Example 3: Isentropic process of ideal gas. I have read the Entropy is defined by: S = Q/TĮntropy is not defined by that equation. Figure 1: With entropy of a closed system naturally increasing, this means that the energy quality will decrease. So at high temperature it pays for the material to hold on to more of its energy, raising the materials entropy. The specific ratio k, varies with temperature, and in isentropic relations above an average k value should be used. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
